Multiscale Assay of Unlabeled Neurite Dynamics Using Phase Imaging with Computational Specificity
M. Kandel E. Kim, Y. Lee, G. Tracy, H. Chung, and G. Popescu
ACS Sensors, April, 21 2021
![]()
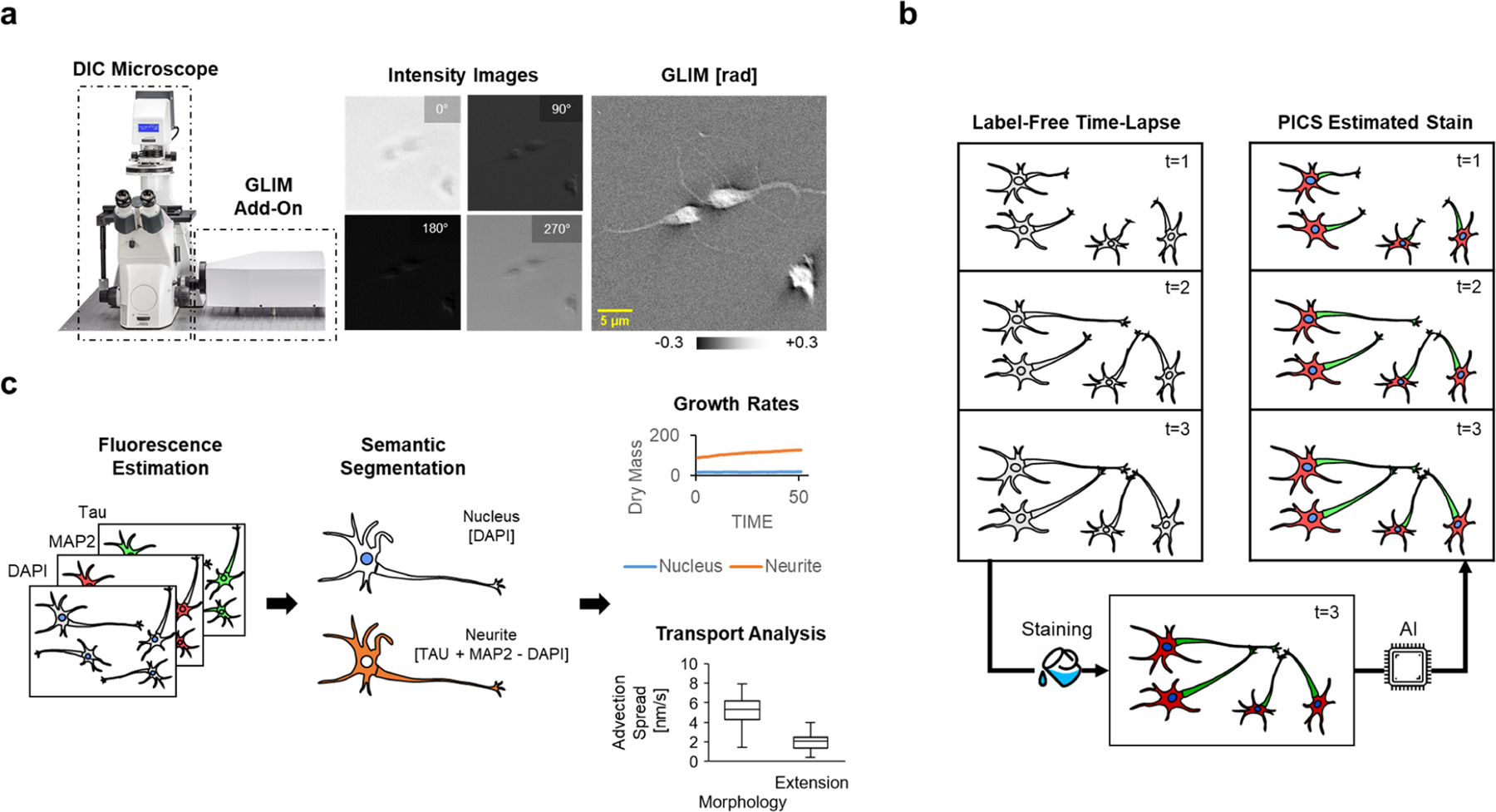
Primary neuronal cultures have been widely used to study neuronal morphology, neurophysiology, neurodegenerative processes, and molecular mechanism of synaptic plasticity underlying learning and memory. However, the unique behavioral properties of neurons make them challenging to study, with phenotypic differences expressed as subtle changes in neuronal arborization rather than easy-to-assay features such as cell count. The need to analyze morphology, growth, and intracellular transport has motivated the development of increasingly sophisticated microscopes and image analysis techniques. Due to its high-contrast, high-specificity output, many assays rely on confocal fluorescence microscopy, genetic methods, or antibody staining techniques. These approaches often limit the ability to measure quantitatively dynamic activity such as intracellular transport and growth. In this work, we describe a method for label-free live-cell cell imaging with antibody staining specificity by estimating the associated fluorescence signals via quantitative phase imaging and deep convolutional neural networks. This computationally inferred fluorescence image is then used to generate a semantic segmentation map, annotating subcellular compartments of live unlabeled neural cultures. These synthetic fluorescence maps were further applied to study the time-lapse development of hippocampal neurons, highlighting the relationships between the cellular dry mass production and the dynamic transport activity within the nucleus and neurites. Our implementation provides a high-throughput strategy to analyze neural network arborization dynamically, with high specificity and without the typical phototoxicity and photobleaching limitations associated with fluorescent markers.
